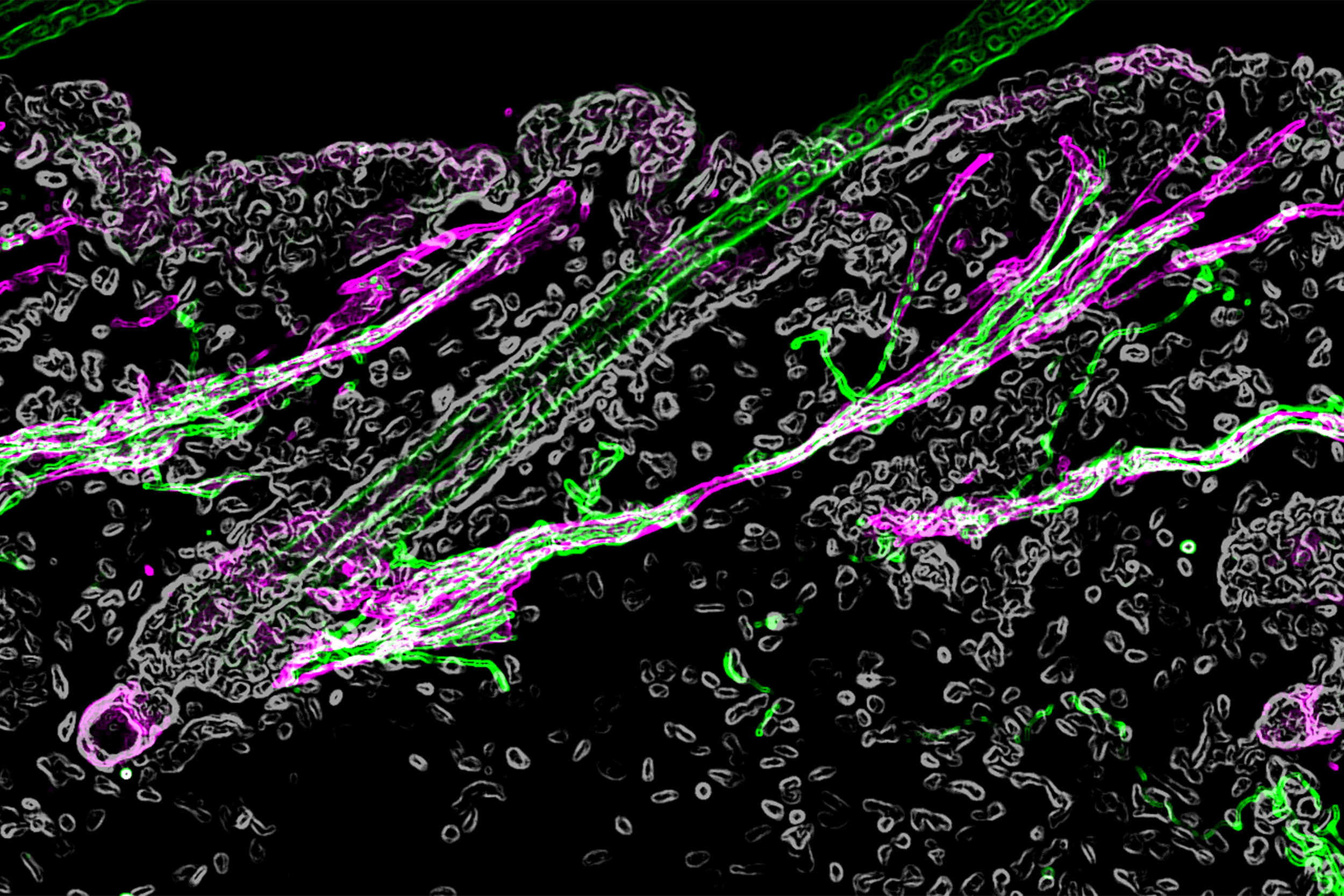
The hair follicle under the microscope, with the sympathetic nerve in green and the muscle in magenta.
Credit: Hsu Laboratory/Harvard University
Getting to the bottom of goosebumps
Harvard scientists find that the same cell types that cause goosebumps are responsible for controlling hair growth
If you’ve ever wondered why we get goosebumps, you’re in good company — so did Charles Darwin, who mused about them in his writings on evolution. Goosebumps might protect animals with thick fur from the cold, but we humans don’t seem to benefit from the reaction much — so why has it been preserved during evolution all this time?
In a new study, Harvard University scientists have discovered the reason: the cell types that cause goosebumps are also important for regulating the stem cells that regenerate the hair follicle and hair. Underneath the skin, the muscle that contracts to create goosebumps is necessary to bridge the sympathetic nerve’s connection to hair follicle stem cells. The sympathetic nerve reacts to cold by contracting the muscle and causing goosebumps in the short term, and by driving hair follicle stem cell activation and new hair growth over the long term.
Published in the journal Cell, these findings in mice give researchers a better understanding of how different cell types interact to link stem cell activity with changes in the outside environment.
“We have always been interested in understanding how stem cell behaviors are regulated by external stimuli,” said Ya-Chieh Hsu, the Alvin and Esta Star Associate Professor of Stem Cell and Regenerative Biology, who led the study in collaboration with Professor Sung-Jan Lin of National Taiwan University.
“The skin is a fascinating system: it has multiple stem cells surrounded by diverse cell types, and is located at the interface between our body and the outside world. Therefore, its stem cells could potentially respond to a diverse array of stimuli — from the niche, the whole body, or even the outside environment,” explained Hsu. “In this study, we identify an interesting dual-component niche that not only regulates the stem cells under steady state, but also modulates stem cell behaviors according to temperature changes outside.”
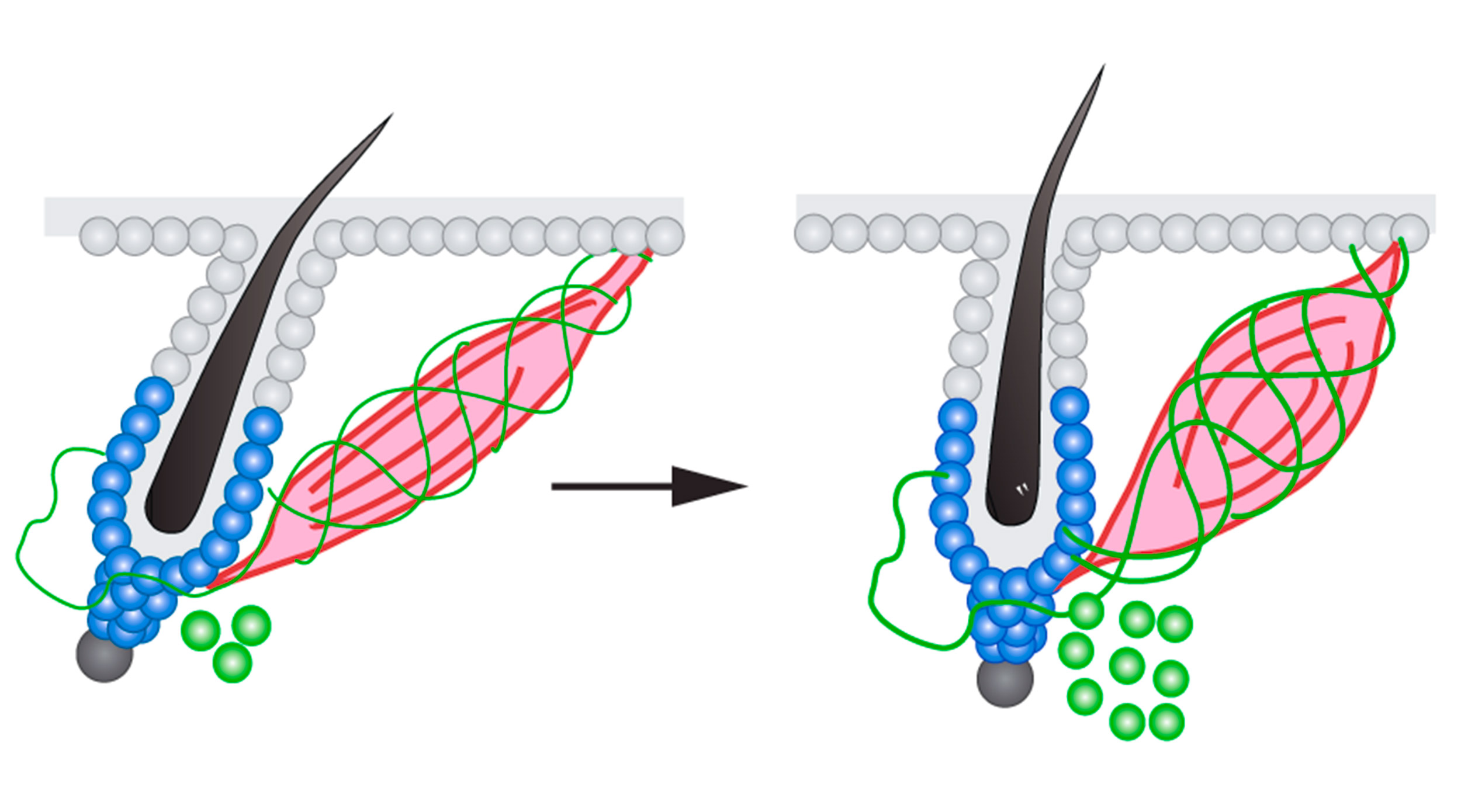
A system for regulating hair growth
Many organs are made of three types of tissue: epithelium, mesenchyme, and nerve. In the skin, these three lineages are organized in a special arrangement. The sympathetic nerve, part of our nervous system that controls body homeostasis and our responses to external stimuli, connects with a tiny smooth muscle in the mesenchyme. This smooth muscle in turn connects to hair follicle stem cells, a type of epithelial stem cell critical for regenerating the hair follicle as well as repairing wounds.
The connection between the sympathetic nerve and the muscle has been well known, since they are the cellular basis behind goosebumps: the cold triggers sympathetic neurons to send a nerve signal, and the muscle reacts by contracting and causing the hair to stand on end. However, when examining the skin under extremely high resolution using electron microscopy, the researchers found that the sympathetic nerve not only associated with the muscle, but also formed a direct connection to the hair follicle stem cells. In fact, the nerve fibers wrapped around the hair follicle stem cells like a ribbon.
“We could really see at an ultrastructure level how the nerve and the stem cell interact. Neurons tend to regulate excitable cells, like other neurons or muscle with synapses. But we were surprised to find that they form similar synapse-like structures with an epithelial stem cell, which is not a very typical target for neurons,” Hsu said.
Next, the researchers confirmed that the nerve indeed targeted the stem cells. The sympathetic nervous system is normally activated at a constant low level to maintain body homeostasis, and the researchers found that this low level of nerve activity maintained the stem cells in a poised state ready for regeneration. Under prolonged cold, the nerve was activated at a much higher level and more neurotransmitters were released, causing the stem cells to activate quickly, regenerate the hair follicle, and grow new hair.
The sympathetic nerve reacts to cold by contracting the muscle and causing goosebumps in the short term, and by driving hair follicle stem cell activation and new hair growth over the long term.
The researchers also investigated what maintained the nerve connections to the hair follicle stem cells. When they removed the muscle connected to the hair follicle, the sympathetic nerve retracted and the nerve connection to the hair follicle stem cells was lost, showing that the muscle was a necessary structural support to bridge the sympathetic nerve to the hair follicle.
How the system develops
In addition to studying the hair follicle in its fully formed state, the researchers investigated how the system initially develops — how the muscle and nerve reach the hair follicle in the first place.
“We discovered that the signal comes from the developing hair follicle itself. It secretes a protein that regulates the formation of the smooth muscle, which then attracts the sympathetic nerve. Then in the adult, the interaction turns around, with the nerve and muscle together regulating the hair follicle stem cells to regenerate the new hair follicle. It’s closing the whole circle — the developing hair follicle is establishing its own niche,” said Yulia Shwartz, a postdoctoral fellow in the Hsu lab. She was a co-first author of the study, along with Meryem Gonzalez-Celeiro, a graduate student in the Hsu Lab, and Chih-Lung Chen, a postdoctoral fellow in the Lin lab.
Responding to the environment
With these experiments, the researchers identified a two-component system that regulates hair follicle stem cells. The nerve is the signaling component that activates the stem cells through neurotransmitters, while the muscle is the structural component that allows the nerve fibers to directly connect with hair follicle stem cells.
“You can regulate hair follicle stem cells in so many different ways, and they are wonderful models to study tissue regeneration,” Shwartz said. “This particular reaction is helpful for coupling tissue regeneration with changes in the outside world, such as temperature. It’s a two-layer response: goosebumps are a quick way to provide some sort of relief in the short term. But when the cold lasts, this becomes a nice mechanism for the stem cells to know it’s maybe time to regenerate new hair coat.”
In the future, the researchers will further explore how the external environment might influence the stem cells in the skin, both under homeostasis and in repair situations such as wound healing.
“We live in a constantly changing environment. Since the skin is always in contact with the outside world, it gives us a chance to study what mechanisms stem cells in our body use to integrate tissue production with changing demands, which is essential for organisms to thrive in this dynamic world,” Hsu said.
This study was supported by the New York Stem Cell Foundation, Smith Family Foundation Odyssey Award, Pew Charitable Trusts, Harvard NeuroDiscovery Center, Harvard Stem Cell Institute, Harvard Medical School Dean’s Innovation Grant, American Cancer Society, National Institutes of Health, Taiwan Ministry of Science and Technology, and National Taiwan University Hospital.




