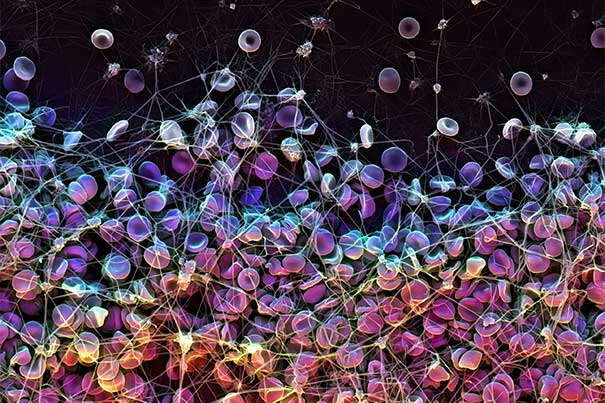
This scanning electron microscope (SEM) image shows how red blood cells coagulate to form a blood clot, which is a common and life-threatening risk associated with the use of implanted medical devices.
Credit: Harvard’s Wyss Institute
SLIPS inspires second generation
Coating repels blood and bacteria, offering lifesaving measures
Most any medical device implanted in the body — or one that comes into contact with flowing blood, such as a dialysis machine — may also present two critical challenges for the patient: blood clotting and bacterial infection.
In a study reported in Nature Biotechnology, a team of Harvard scientists and engineers has developed a new surface coating for medical devices using materials already approved by the Food and Drug Administration (FDA). The researchers noted that the coating repelled blood from more than 20 medically relevant substrates (glass, plastic, and metal) and also suppressed biofilm formation.
The team also implanted medical-grade tubing and catheters coated with the material in large blood vessels in pigs. The coating prevented blood from clotting for at least eight hours without the use of blood thinners like heparin, which can cause side effects, such as excessive bleeding.
“Devising a way to prevent blood clotting without using anticoagulants is one of the holy grails in medicine,” said Don Ingber, director of Harvard’s Wyss Institute for Biologically Inspired Engineering and senior author of the study. Ingber is also the Judah Folkman Professor of Vascular Biology at Harvard Medical School and Boston Children’s Hospital, as well as professor of bioengineering at Harvard’s School of Engineering and Applied Sciences (SEAS).
The idea for the coating evolved from SLIPS (slippery-liquid-infused porous surfaces), a pioneering surface technology developed by co-author Joanna Aizenberg, who is a Wyss Institute core faculty member and the Amy Smith Berylson Professor of Materials Science at SEAS.
SLIPS was inspired by the slippery surface of the carnivorous pitcher plant. It repels nearly any material it contacts, and the liquid layer on its surface provides a barrier against everything from ice to crude oil and blood.
“Traditional SLIPS uses porous, textured surface substrates to immobilize the liquid layer, whereas medical surfaces are mostly flat and smooth — so we further adapted our approach by capitalizing on the natural roughness of chemically modified surfaces of medical devices,” said Aizenberg, who leads the Wyss Institute’s adaptive materials group. “This is yet another incarnation of the highly customizable SLIPS platform that can be designed to create slippery, nonadhesive surfaces on any material.”
In a two-step surface-coating process, the Wyss team chemically attached a monolayer of perfluorocarbon, which is similar to Teflon, then added a layer of liquid perfluorocarbon. The team calls the tethered perfluorocarbon plus the liquid layer a “tethered-liquid perfluorocarbon” surface, or TLP.
The TLP coating achieved the following results:
- TLP-treated medical tubing stored for more than a year under normal temperature and humidity conditions still prevented clot formation.
- The TLP surface remained stable under the full range of clinically relevant physiological shear stresses, or rates of blood flow seen in catheters and central lines, all the way up to dialysis machines.
- The TLP coating repelled the components of blood that cause clotting (fibrin and platelets).
- When bacteria called Pseudomonas aeruginosa were grown in TLP-coated medical tubing for more than six weeks, less than one in a billion bacteria were able to adhere.
The researchers even tested a TLP-coated surface with a gecko — the superstar of sticking whose footpads contain thousands of hairlike structures with tremendous adhesive strength. The gecko was unable to hold on.
“We were wonderfully surprised by how well the TLP coating worked, particularly in vivo without heparin,” said one of the co-lead authors, Anna Waterhouse, a Wyss Institute postdoctoral fellow. “Usually the blood will start to clot within an hour in the extracorporeal circuit, so our experiments really demonstrate the clinical relevance of this new coating.”
The project was funded by the Defense Advanced Research Projects Agency (DARPA) and the Wyss Institute.




