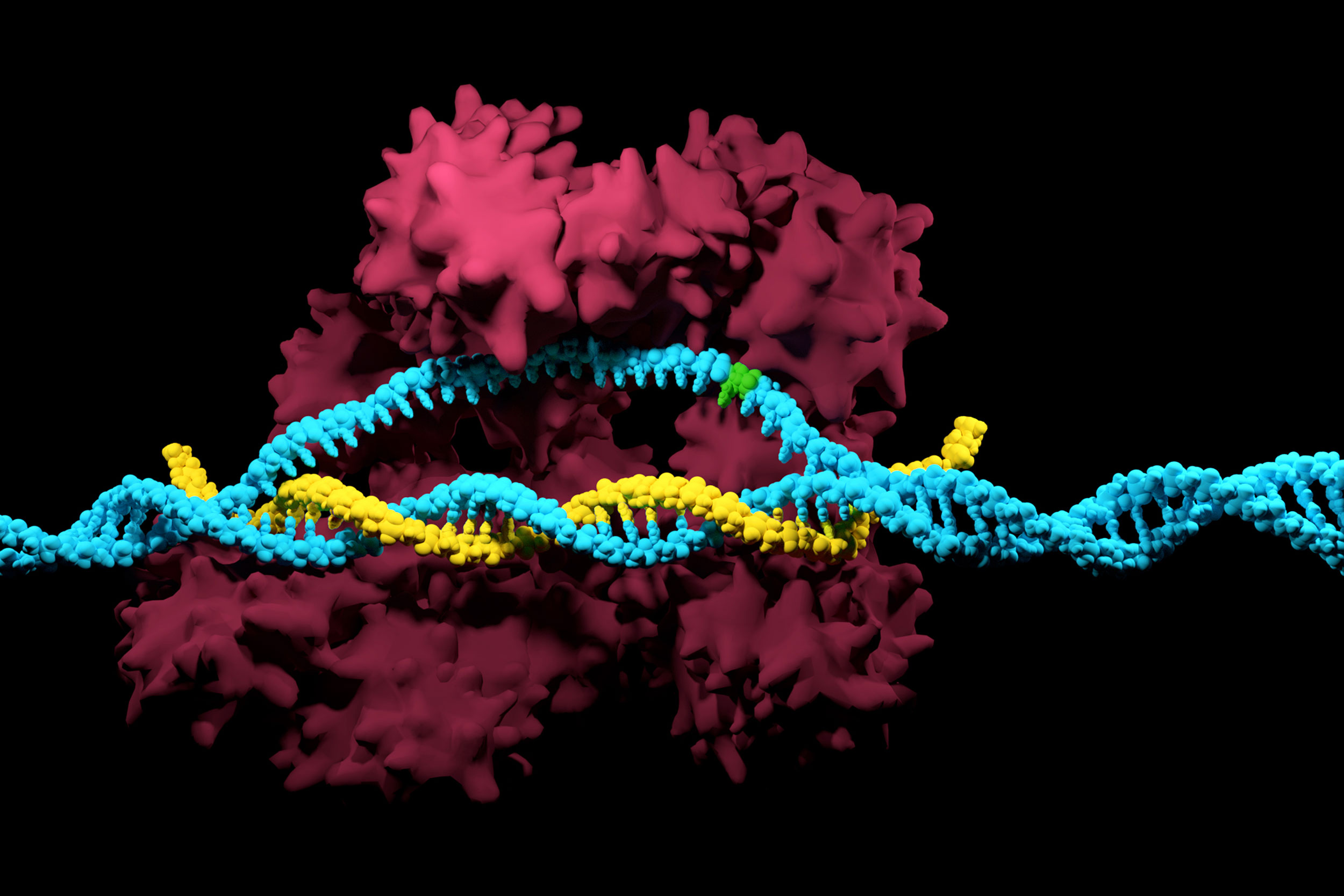
A Wyss Institute team devised a CRISPR/Cas9-based mutation-prevention system that is capable of discriminating a single nucleotide variation (indicated here in green) in the DNA code (in blue) to readily remove newly occurring disease-associated mutations.
Credit: iStock/Meletios Verras
Keeping the genetic code clean
A CRISPR/Cas9 mutation prevention system could help prevent and fight disease in the future
Many life-threatening diseases are caused or exacerbated by a change of just one nucleotide building block in the universal genetic DNA code. Such “point mutations” can turn a single cell in the human body into a cancerous cell that grows into a tumor, or turn antibiotic-sensitive into antibiotic-resistant bacteria that cause untreatable infections. In an ideal world, clinicians would be able to remove cells with such deleterious point mutations right after they were created to fight diseases much more effectively.
A research team at the Wyss Institute for Biologically Inspired Engineering at Harvard University reports now in the Proceedings of the National Academy of Sciences (PNAS) that it has accomplished the first step toward this goal by converting the CRISPR/Cas9 genome-engineering system into a genome-surveillance tool. The researchers, led by Wyss Institute core faculty members George Church and James Collins, developed an in vivo mutation prevention method that enables the DNA-cleaving Cas9 enzyme to discriminate between genomic target sites differing by a single nucleotide and cut only the unwanted one. In proof-of-concept studies performed in bacterial E. coli strains grown in culture or the mouse gastrointestinal tract, the approach can prevent the survival of antibiotic-resistant variants.
The Cas9 enzyme is guided to its genomic target sequence by a small guide RNA with a complementary sequence. Once brought into position on the gene of interest, Cas9 acts like a molecular pair of scissors, cutting the target sequence at a defined site. If repaired, this deliberately introduced damage allows biologists to edit the genome of cells, but if left unrepaired, it will cause cells to die. Yet despite the system’s effectiveness in finding and cutting target sequences in the genomes of many organisms, unspecific activity that lets Cas9 randomly cut at secondary, not completely identical sites still poses a problem to genome engineers. This also means that a target sequence carrying an undesired point mutation often cannot be sufficiently discriminated by Cas9 from its normal counterpart and selectively removed. Even designer Cas9 enzymes engineered to be more specific so far are not able to solve this problem.
“We are now able to prevent often-occurring mutations that confer antibiotic resistance and ask which other genetic changes could lead to the same result.”
James Collins
“By focusing instead on guide RNA features, our approach dramatically enhances Cas9’s specificity up to a level where single nucleotide polymorphisms can be clearly distinguished and unwanted genetic variants erased,” said Church, who also is professor of genetics at Harvard Medical School (HMS) and of health sciences and technology at Harvard and the Massachusetts Institute of Technology. “Our method opens up an entirely new way to think about disease prevention in the future.”
Previous studies had shown that, paradoxically, certain mismatches between a guide RNA and its target sequence did not affect Cas9’s ability to cleave a specific site in the DNA. “We hypothesized that for a given pair of targets that differ by a single point mutation, a set of mismatches could be identified in the guide RNA that would eliminate Cas9’s activity on the normal sequence while maintaining robust activity on the one with a deleterious point mutation. This would stop a cell with a mutation cold in its tracks right after it is born,” said Alejandro Chavez, a first and co-corresponding author on the study, who as a postdoctoral fellow at HMS was co-mentored by Church and Collins and is now assistant professor at Columbia University.
To develop their approach, the team leveraged known point mutations that occur in bacterial enzymes, providing pathogens with resistance to antibiotics. By focusing on several of these mutations and screening through guide RNA variants with different mismatch combinations, they were able to identify specific guide RNAs that stimulated Cas9 activity toward the mutated gene sequences but left the normal counterpart untouched.
“The mutation-prevention system maintained antibiotic sensitivity not only in E. coli strains cultured in standard laboratory conditions, but also in bacteria that were used to colonize the gastrointestinal tract of gnotobiotic mice. These multiday mouse experiments involved sustained antibiotic dosing and demonstrated that the system is robust even when subjected to potentially significant environmental pressures,” said co-first author Benjamin Pruitt, a former staff research scientist at the Wyss Institute and now lead research and development engineer at ReadCoor Inc.
Besides its potential for future disease prevention, the researchers think their mutation-prevention system could more immediately be used to help the biotech industry protect large-scale cultures from acquiring mutations that render them unproductive or prone to contaminations, and to study microbial evolution.
“This strategy gives us the opportunity to study evolutionary mechanisms in microorganisms. For example, we are now able to prevent often-occurring mutations that confer antibiotic resistance and ask which other genetic changes could lead to the same result. This may improve our understanding of resistance mechanisms and potentially offer new therapeutic entry points,” said Collins, who is also the Termeer Professor of Medical Engineering & Science at MIT, and a professor of biological engineering there.
“This collaborative effort at the Wyss Institute has resulted in an entirely new innovation in the CRISPR/Cas9 field, one that could lead to entirely new ways to prevent and treat a broad range of diseases,” said Wyss Institute Director Donald Ingber, who is also the Judah Folkman Professor of Vascular Biology at HMS and the Vascular Biology Program at Boston Children’s Hospital, as well as professor of bioengineering at the Harvard John A. Paulson School of Engineering and Applied Sciences.
Additional authors on the study are Wyss Institute researchers Rebecca Shapiro, a former member of Collins’ group and now assistant professor at the University of Guelph, Canada; and Marcelle Tuttle, Ryan Cecchi, Jordan Winston, Brian Turczyk, and Michael Tung, who are past and present members of Church’s group.
The study was funded by Harvard’s Wyss Institute for Biologically Inspired Engineering, the National Institutes of Health, the National Cancer Institute, the Burroughs Wellcome Fund, the Defense Threat Reduction Agency, and the Paul G. Allen Frontiers Group.








