Wyss Institute receives grant from Cystic Fibrosis Foundation
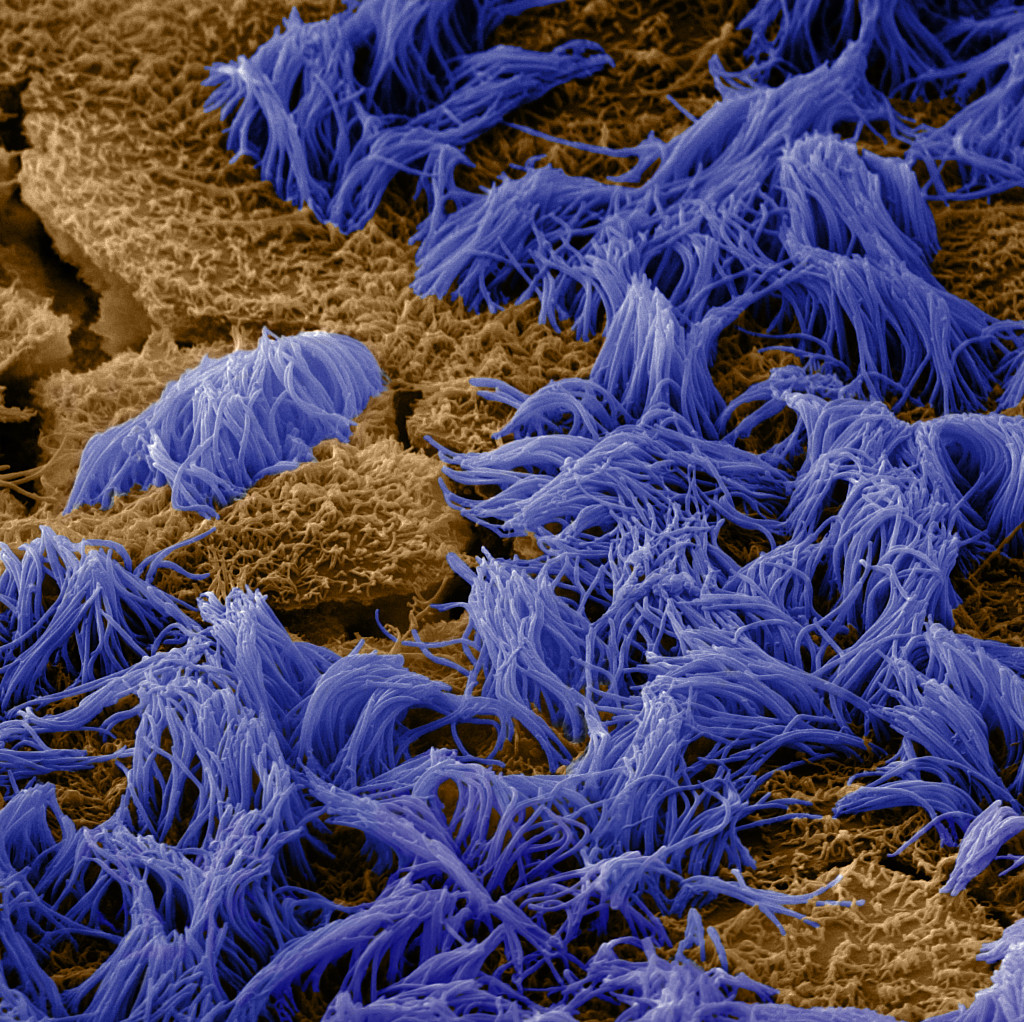
Lung cells are covered with fine, hair-like projections called cilia (purple), which help sweep mucus and debris out of the airways. Credit: Wyss Institute at Harvard University
A new grant from the Cystic Fibrosis Foundation (CFF) to the Wyss Institute at Harvard University aims to improve the survival of patients with cystic fibrosis (CF) by funding the development of a human Airway Chip model of a CF lung that can be used to study the bacterial infections and inflammation that often lead to deadly respiratory failure in patients with CF.
“This tool will enable researchers to analyze the molecular mechanisms that underlie the lung’s response to pathogens in patients with CF, and provide a human-relevant model in which to test potential CF therapeutics,” said Wyss Founding Director Donald Ingber, who is also the Judah Folkman Professor of Vascular Biology at HMS and the Vascular Biology Program at Boston Children’s Hospital, as well as Professor of Bioengineering at Harvard SEAS.
CF, caused by a recessive genetic mutation in the CFTR gene, dramatically shortens patients’ lifespans, largely due to chronic respiratory complications. The CFTR protein functions as an ion channel, helping to maintain a thin layer of water on the inner surface of the lung that allows tiny hairs called “cilia” on the lung cells to sweep back and forth, clearing mucus out of the airways. When the CFTR protein is disrupted, the water layer breaks down, preventing the cilia from clearing mucus and creating a breeding ground for bacteria that can become highly infectious and resist the body’s immune system.
“Organ Chips allow us to create experimental conditions — including co-culturing human cells with living microbes — within an in vitro model that behaves like a human organ, making them the perfect vehicle for studying how bacterial pathogens interact with their hosts at the cellular level,” said Rachelle Prantil-Baun, a senior staff scientist at the Wyss Institute who is helping lead the CF Airway Chip project.
The Wyss Institute has already developed Organ Chips mimicking various human organs, all of which display human organ-level functions. “Because all of the parameters in the Organ Chip system can be varied individually, it offers a novel ‘synthetic biology’ approach to study how cellular, molecular, chemical, and physical cues influence human tissue development and disease,” said Ratnakar Potla, a postdoctoral fellow at the Wyss Institute who is the technical lead of the CF Airway Chip project.
This project will use a modified version of the Wyss’ Lung Small Airway Chip, with its epithelial channel lined with CFTR-deficient human airway cells from CF patients and its endothelial channel with healthy blood vessel cells (both provided by the CFF), and analyze the function of those organ models both with and without bacteria. The CF Airway Chips will also have integrated electrodes to quantify the movement of ions through the CFTR protein channels in real time.
The team hopes to gain a better understanding of how pathogens influence CF airway structure and function, and identify novel ways to treat chronic inflammation in patients with CF.
To learn more about cystic fibrosis or the Cystic Fibrosis Foundation, please visit www.cff.org.




