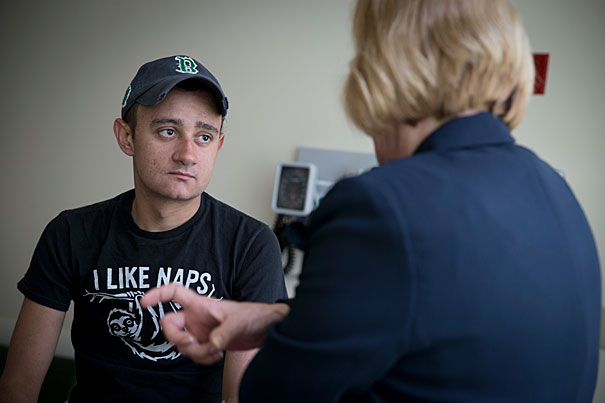
Brenden Whittaker, 22, of Ohio, is a patient with a rare genetic immune disease in a Children’s Hospital gene therapy trial. Pediatric Oncology RN, Brenda MacKinnon and Whittaker discuss his progress.
Rose Lincoln/Harvard Staff Photographer
Finally, hope for a young patient
Gene therapy trial at Boston Children’s shows promise against devastating immune disorder
Brenden Whittaker woke in his room at Nationwide Children’s Hospital in Columbus, Ohio, drenched in sweat.
It was 3 a.m., and his mother, Becky Whittaker, watched as the nurse tried to take his temperature with an electronic thermometer, then left to fetch an old-style mercury one that registered higher.
It read 105.
“I really thought, ‘We’re going to lose him to this disease,’” Becky recalled. “‘He’s 16 years old, and he’s going to be one of the statistics.’ … He was so, so sick.”
The nurse helped Brenden change out of his sweat-soaked clothes before leaving the two alone in the dark of that too-early morning. Becky settled into the rocking chair next to the bed.
“He started talking to me about planning his funeral, where he wanted to be buried, what kind of funeral he wanted,” Becky said. “He said, ‘Mom, if you think I’m going to die, will you tell me? I’m begging you — you have to tell me if you think I’m going to die, if I’m not going to get better.’
“I looked at him and I said, ‘It’s going to rip me apart, but, yes, I’ll tell you. … I don’t think we’re there now.’”
That night in the fall of 2009 was a new low in the course of Brenden’s disease, a rare immune disorder called chronic granulomatous disease, or CGD. The condition affects a key component of the immune system, a type of white blood cell called a neutrophil that engulfs and kills invading bacteria and fungi.
Since his diagnosis as a baby, Brenden had taken regular preventive antibiotics to fight off infections before they started, gotten thrice-weekly interferon shots to boost his weakened immune system, and avoided activities that might bring him into contact with troublesome microbes. Despite that care, he’d been hospitalized repeatedly for surgeries to drain abscesses and for intravenous antibiotics to fight infections.
“[Over the years], I’ve had hundreds of procedures,” Brenden said. “I’ve had lung biopsies, bronchoscopies, endoscopies, had ingrown toenails removed. I’ve had all kinds of stuff like that.”
Despite his fears in the hospital that night, Brenden recovered, but it cost him the infected portion of his lung, which doctors removed, and his junior year in high school. Even then, the recovery wasn’t permanent. His next downward spiral began in 2014, and in April 2015 he had the left lobe of his liver removed, before spending most of August at Nationwide Children’s to treat another lung infection.
Today, though, Brenden’s lungs are clear and, for the first time in his life, roughly half of his neutrophils are functioning — enough, doctors say, to keep him well. Now 23, he is working again, part time at a golf course near his home in Ohio, and cautiously looking forward to college in 2017 — if his health holds out.
The sea change came after Brenden received an experimental gene therapy treatment in December at the Dana-Farber/Boston Children’s Cancer and Blood Disorders Center, a collaboration of Boston Children’s Hospital and the Dana-Farber Cancer Institute. This phase I trial, intended to test the procedure’s safety, is being conducted at three centers around the country and is led in Boston by David Williams, the Leland Fikes Professor of Pediatrics at Harvard Medical School (HMS), chief of hematology/oncology and director of clinical and translational research at Boston Children’s, and president of the Dana-Farber/Boston Children’s Cancer and Blood Disorders Center. Williams has worked in the gene-therapy field since he was a postdoctoral fellow at Massachusetts Institute of Technology in the early ’80s.
“I’m thrilled we were able to get the trial opened so quickly and enroll our first patient,” Williams said. “I’m very thrilled everything has gone well so far.”
The trial is designed to progress in stages, so Brenden’s improved health cleared the way for a second patient to begin the same treatment, this time at the National Institutes of Health in Bethesda, Maryland. If all goes well with both patients, a third will undergo the procedure at the University of California at Los Angeles, where Donald Kohn is the chief investigator for the overall study. These three early patients will be closely monitored and, if there are no complications, more will be treated with the aim of moving to a broader, phase II trial. The ultimate goal is to create a new CGD treatment, which would be among the vanguard of new therapies based on altering a patient’s genetic code.
Where’s the bleach?
When chronic granulomatous disease was first recognized in 1950, little could be done to control the infections that plagued its sufferers, who rarely lived past 10.
“The disease was first called ‘fatal granulomatous disease,’ which gave a good impression of what happened. Prior to good antibiotics … the disease was fatal,” said Stuart Orkin, the David G. Nathan Professor of Pediatrics at HMS and who is affiliated with the Dana-Farber/Boston Children’s Cancer and Blood Disorders Center. Orkin has studied the genetics of CGD for decades.
Over time, doctors learned the lesson they would apply in Brenden’s case: that the way to keep someone with CGD alive is not to let them get sick in the first place.
Though modern antibiotics have made CGD a manageable disease, control can be imperfect. Life with CGD is marked by repeated infections, large and small, and regular inpatient hospital stays to fight the worst flare-ups. Patients develop granulomas — the body’s efforts to wall off infections it cannot defeat — which cause problems of their own, blocking digestive tracts and other critical passages.
CGD affects about 1,500 people nationwide and has two major variants. A patient can inherit both copies of a defective gene from his or her parents or, as in Brenden’s case, because the defective gene is carried on a mother’s X chromosome, and boys have only one X chromosome, only one copy can cause disease.
In the body, the defective gene upsets the killing chemistry that occurs inside healthy neutrophils. These white blood cells engulf bacteria or fungi, triggering a series of chemical reactions that ultimately produce hypochlorite, the active ingredient in common household bleach. The hypochlorite, produced in tiny quantities, kills the engulfed invader.
In the 1960s, a pediatric hematology/oncology fellow at Boston Children’s named Robert Baehner devised the first diagnostic test for CGD. Baehner went on to become a professor at Indiana University School of Medicine, where part of his continued impact on the field was training the leader of Brenden’s current gene therapy trial, Williams.
“I’m here today because I had such a wonderful mentoring experience with Dr. Baehner when I was a medical student,” Williams said. “I hadn’t done research before that, so this completely changed my career.”
Baehner also returned to Children’s years later on sabbatical to work with Orkin, bringing with him a tissue sample from a CGD sufferer.
In 1986, Orkin, working with colleagues searching for the muscular dystrophy gene — thought to be near CGD’s gene — used that sample to identify the CGD gene, pioneering a technique called positional cloning.
“Working together [with collaborators], we did a number of … things, using some logic, a little luck, good reagents,” Orkin said. “It was very challenging. The extent of what we knew about the genome … was like the Stone Age compared to today.”
Baby blues
Brenden Whittaker’s CGD revealed itself only gradually.
After a family trip to the beach at age 1, Brenden developed a grape-sized swelling on the right side of his face. Routine antibiotics didn’t work, and a mumps test came back negative, so the doctor drained the infection and sent a sample to the lab for testing.
Results brought more questions than answers. The infection was caused by a bacterium common in dirty water, Serratia marcescens, but one that a healthy immune system should be able to handle. That fall, when Brenden developed a strange, acne-like bump on his chin also caused by Serratia marcescens, it was apparent there was an ongoing problem.
The doctor quizzed Becky about her family history, looking for signs of immune dysfunction: Had anyone died young, died of infection?
“At first you’re like, ‘Well, no way this could be it. I can’t think of any child in my family with any issues or any problems like this,’” Becky said. “I grew up on a farm, shoveling out stalls with manure and whatnot, and I was never sick. But half of my white cells work.”
A DNA test of Becky’s blood was positive for CGD. So she and Brenden traveled to the National Institutes of Health, and saw Harry Malech, a CGD expert. He counseled them about Brenden’s careful way forward.
“It’s a really hard disease for a boy to have because, statistically speaking, more boys would prefer to be dirty than not,” Becky said.
Despite periodic trips to the hospital, Brenden was able to live an active life. His mother’s work as a transplant nurse ensured that someone knowledgeable about health and the health care system watched over him.
He played soccer and ice hockey, avoiding baseball, football, and their bacteria-laden dirt, dust, and grass. He backpacked with the Boy Scouts, packing all his water to avoid questionable sources on the trail. He demonstrated canoe and kayak skills in a pool’s clean waters.
But infection and illness were never far away. Pneumonia forced him to start kindergarten late and with an IV for antibiotics. In second grade, his small intestine was almost blocked by a granuloma, which stubbornly persisted until a cocktail of antibiotics, high-dose steroids, and other drugs did the trick. In sixth grade, he was back in the hospital with an ankle infection.

But it was during 11th grade that things really went south. The “rip-roaring infection,” as Becky described it, settled in his lungs in September and held on. For months, the Nocardia bacteria shrugged off antibiotics until, finally, in March, surgeons removed the upper and part of the middle lobe of his right lung.
“The doctor who did the surgery brought the tissue out into the waiting room and showed it to us. He had developed a big granuloma around the Nocardia infection so the antibiotics were not going to get to it. They weren’t ever going to take care of the infection with no white cells to help them work,” Becky said. “[The year] 2009–10 was a terrible, terrible period of time. … You can’t just keep chopping away essential body parts when new infections occur.”
Brenden recovered, but had missed so much school that he repeated his junior year. By late 2014, he had graduated and started at a local community college when he got sick again. He would fight infections through much of 2015.
“I probably spent nine of 12 months of the year in the hospital. I had a granuloma in my urethra in January. I had the liver stuff kind of February through May. I had some recurring pneumonia in my right lung, or, actually, in both of my lungs,” Brenden said. “I spent the Fourth of July in the hospital and was discharged before my birthday in July. I was in the hospital by Aug. 2. I had a lung biopsy that started to bleed. I spent a week in the ICU in August, then I was admitted to the regular inpatient floor for three weeks following that, so I was in the hospital all of August. They assumed it was a fungal infection in my lungs. They never figured out what it was.”
Brenden’s doctor at Nationwide Children’s told them what had become apparent: Brenden was getting too many infections, having too many surgeries and, with so much antibiotic use, risking drug-resistant infections. They needed to explore other options.
Bone-marrow transplants can provide a source of healthy immune cells for CGD sufferers, but tests of family members and unrelated donors turned up no good matches. Finally, a specialist who had heard of Williams’ gene-therapy trial at Boston Children’s thought Brenden might be a good candidate.
“I had been so sick last year, I was willing to do anything not to be that sick anymore,” Brenden said.
Gene therapy
After a sputtering start, gene therapy has gathered momentum. Its promise was illustrated in 1990 when a 4-year-old girl named Ashanti DeSilva became the world’s first successful gene-therapy patient. The treatment did not cure the near-total immune dysfunction that Ashanti suffered, but nonetheless it pulled her out of danger, allowed her to enroll in school, and illustrated the merit of treating genetic diseases at their most basic level.
Progress stalled in 1999, when 18-year-old Jesse Gelsinger became the first person to die in a gene-therapy trial. Gelsinger, who had a genetic liver disease, had a massive immune reaction to the virus, called a vector, that was used to shuttle the corrected gene for his condition into his DNA.
“Gene therapy has had a number of setbacks along the way,” Orkin said. “Those setbacks probably have delayed things somewhat. … Each one of those steps takes five years to recover from … so it’s taken a long time.”
In 2008, researchers successfully treated Leber’s congenital amaurosis, which causes blindness. In 2010, a French patient was treated for beta-thalassemia major, a blood disease. In 2013 and 2014, children were treated for ADA-SCID, or “bubble boy syndrome.” Other trials have targeted hemophilia, the inherited eye disease choroideremia, HIV, sickle cell disease, and cancer.
Venture capital firms have taken notice of the progress, and money has flowed into gene therapy-based startups, a good sign, according to Williams — who, with colleagues at Boston Children’s and three other academic institutions in the United States and United Kingdom, recently started a gene therapy company — since such firms expect short-term returns that flow from clinical success.
“I think things are progressing,” Williams said. “You get a sense of that by the number of trials that are open, and you get a sense of that by the number of new companies that are starting.”
Though Brenden later admitted some apprehension at being the first to undergo the procedure, he agreed to the trial and traveled to Boston last November for screening tests. Physicians also extracted blood stem cells to freeze as backup in case the procedure went awry.
In early December, Brenden returned so doctors could take more blood stem cells, this time to be engineered for the trial at a cell-manufacturing facility run by Dana-Farber.
There, the cells were isolated and mixed with a virus containing a functioning copy of Brenden’s malfunctioning gene. The virus inserted the working gene into the cells’ DNA, creating a population of Brenden’s own stem cells with an extra gene. The hope was that these stem cells, which would be infused back into Brenden later that month, would develop into properly functioning white blood cells.
On Dec. 13, Brenden returned to Boston and the next day began a three-day, six-dose course of chemotherapy, which killed cells in his bone marrow to clear space for the new, engineered cells to take hold. On the 18th, the corrected stem cells were infused into his blood to begin their migration to the marrow.
Brenden doesn’t remember much of his nearly month-long stay, little of Christmas or college football’s Bowl season. The nausea after chemo was unforgettable, though, as were the sores in his mouth and the hair that was falling out.
Ohio State beat Notre Dame in the Fiesta Bowl. “I have been told I watched the game in my hospital room, and I asked a few days later when they were supposed to play,” Brenden said. “I didn’t know it was the new year for a week.”
He was discharged in early January, but stayed in Boston for outpatient checkups until the end of the month. At home in Ohio, he had three more months of forced inactivity while his immune system rebooted.
“I was definitely apprehensive about it before,” Brenden said. “Looking back now, I’m glad that I did it.”
In the months since, Brenden has resumed his old job at the local golf course, gone running, and played some golf himself. He’s also gotten regular checkups and taken periodic trips back to Boston.
The results have been encouraging, with one spring follow-up test showing nearly half of Brenden’s neutrophils working properly. That level is well above the trial’s target of 10 percent and enough for researchers to go ahead with a second patient.
“At 10 percent, our expectation is it will provide the person protection to make them essentially normal,” Williams said. “We’re not looking for partial protection, we’re looking for full protection. We think that will be the case.”
No promises
Gene therapy’s ultimate goal is to make a one-time fix, correct a genetic disease’s underlying cause, and let people get on with their lives.
In this case, however, Williams said it’s too early to talk about a widespread CGD cure. Though promising, this therapy is at the proof-of-concept stage. If it succeeds in additional trials, initially it would be reserved for the sickest patients — like Brenden — who are having difficulty managing their illness and aren’t good candidates for bone-marrow transplants. If the therapy proves itself over time, its use could expand to aid patients before serious complications develop.
“It’s very, very early, of course,” Williams said. “It’ll be an evolution with time, of a broadening indication, if everything goes well.”
For Brenden, if all goes well he’ll be back in school in January. He plans to return to community college with an eye to transferring to Ohio State, and perhaps medical school after that.
For his mother, each encouraging neutrophil count is exciting, but experience has taught her caution.
“I don’t think Brenden and I are under any illusions that this is a cure,” Becky said. “People ask what I’ll do with the time [if Brenden stays healthy]. There are things I’d like to do, but I’m cautious about starting them because part of me is wondering when the other shoe is going to drop and what it will be when it does.
“I tried to think about how I’d feel if at some point this isn’t working, the cells reverting to their defective ways. I can’t predict how I’ll feel when that happens, so I just dwell on each measurement and, OK, this is just another period of time when the measurement is good. Let’s just go about what we’re doing. I think that’s just about all we can do.”





