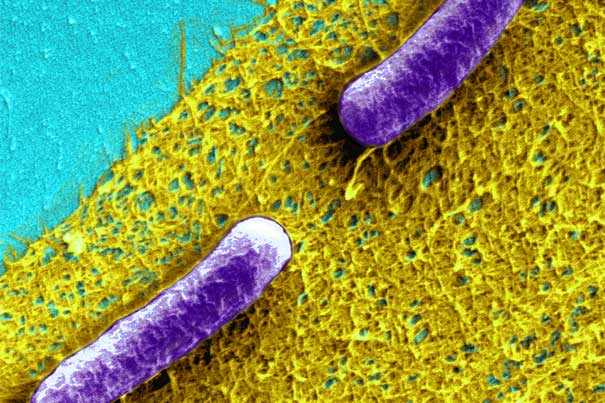
Biofilms are communities of bacteria ensconced in a matrix of slimy, but extremely tough, extracellular material composed of sugars, proteins, genetic material, and more. Associate Professor of Chemical and Biological Engineering Neel Joshi and his team wanted to give them a facelift, and developed a novel protein-engineering system called BIND to do so.
Image courtesy of the Wyss Institute
Recruiting bacteria for innovation
Harvard team lays foundation for using biofilms to create self-healing materials and other technologies
For many people, biofilms conjure up images of slippery stones in streambeds or dirty drains. A team at the Wyss Institute for Biologically Inspired Engineering at Harvard University thinks of them instead as a robust new platform for designer nanomaterials that could help clean polluted rivers, manufacture pharmaceutical products, fabricate new textiles, and more.
Biofilms are communities of bacteria ensconced in a matrix of slimy, but extremely tough, extracellular material composed of sugars, proteins, genetic material, and more. Researchers wanted to give them a facelift, and developed a novel protein-engineering system called BIND to do so. With BIND, which stands for biofilm-integrated nanofiber display, the team said biofilms could become living foundries for large-scale production of biomaterials that can be programmed to provide functions not possible with existing materials. They reported the proof-of-concept today in the journal Nature Communications.
“Most biofilm-related research today focuses on how to get rid of biofilms, but we demonstrate here that we can engineer these super-tough natural materials to perform specific functions, so we may want them around in specific quantities and for specific applications,” said Wyss Institute Core Faculty member Neel Joshi, the study’s senior author. Joshi is also an associate professor of chemical and biological engineering at the Harvard School of Engineering and Applied Sciences (SEAS).
Biofilms also can self-assemble and self-heal. “If they get damaged, they grow right back because they are living tissues,” said lead author Peter Nguyen, a postdoctoral fellow at the Wyss Institute and SEAS.
During biofilm formation, individual bacteria pump out proteins that self-assemble outside the cell, creating tangled networks of fibers that essentially glue the cells together into communities that keep the bacteria safer than they would be on their own.
Interest in biofilm engineering is skyrocketing, and while several other teams have recently developed genetic tools to control biofilm formation, Joshi’s team altered the composition of the extracellular material itself, essentially turning it into a self-replicating production platform to churn out whatever material they wish to produce.
“Until recently, there was not enough cooperation between synthetic biologists and biomaterials researchers to exploit the synthetic potential of biofilms this way. We are trying to bridge that gap,” Joshi said.
The team genetically fuse a protein with a particular desired function — for example, one known to adhere to steel — onto a small protein called CsgA that is already produced by E. coli bacteria. The appended domain goes along for the ride through the natural process by which CsgA is secreted outside the cell, where it self-assembles into super-tough proteins called amyloid nanofibers. These amyloid proteins retain the functionality of the added protein, ensuring the desired function, in this case that the biofilm adheres to steel.
Amyloid proteins traditionally get a bad rap for their role in causing tremendous health challenges such as Alzheimer’s disease, but here their role is fundamental to making BIND robust. The amyloids can spontaneously assemble into fibers that, by weight, are stronger than steel and stiffer than silk.
“We are excited about the versatility of the method, too,” Joshi said. The team demonstrated an ability to fuse 12 different proteins to the CsgA protein, with widely varying sequences and lengths. This means that in principle they can use this technology to display virtually any protein sequence — a significant feature because proteins perform an array of impressive functions, from binding to foreign particles, to carrying out chemical reactions, to transmitting signals, providing structural support, and transporting or storing certain molecules.
Not only can these functions be programmed into the biofilm one at a time, they can be combined to create multifunctional biofilms as well.
The concept of the microbial factory is not a new one, but this is the first time it is being applied to materials, as opposed to soluble molecules such as drugs or fuels. “We are essentially programming the cells to be fabrication plants,” Joshi said. “They don’t just produce a raw material as a building block, they orchestrate the assembly of those blocks into higher-order structures and maintain those structures over time.”
“The foundational work Neel and his team are doing with biofilms offers a glimpse into a much more environmentally sustainable future, where gargantuan factories are reduced to the size of a cell that we can program to manufacture new materials that meet our everyday needs — from textiles to energy and environmental clean-up,” said Wyss Institute Founding Director Don Ingber.
For now, the team has demonstrated the ability to program E. coli biofilms that stick to certain substrates such as steel, and others that can immobilize an array of proteins or promote the templating of silver for construction of nanowires.
This work was primarily funded by the Wyss Institute. The authors also acknowledge support from the National Science Foundation Graduate Research Fellowship and the A*STAR National Science Graduate Fellowship.




