Research promises new paths to treatments, cures
Scientists ask of stem cells: ‘How does it do that?’
HARVARD STEM CELL INSTITUTE
Stem cell videos:
- Dr. Melton researches stem cells
- Dr. Scadden prescribes stem cell therapy
- HSCI hopes to accelerate research
- Dr. Scadden on embryonic stem cells
[default format for video is RealPlayer. For videos in Quicktime format, visit http://www.harvard.edu/multimedia/]
– More stories about stem cells
– Harvard Alumni Association videoconference on stem cells, “Unlocking the promise of stem cells”
– Harvard Stem Cell Institute hosts inaugural symposia (press release 4/23/04)
At DNA’s mysterious command, an embryonic stem cell can somehow become heart, lung, liver, bone, hair, skin, nail, or any other tissue in the body.
First identified in 1998, but hypothesized long before, human embryonic stem cells are the foundation on which the body is built. Researchers have imagined the benefits of harnessing this cell’s power, directing the creation of badly needed tissues and organs to cure human disease, or creating banks of human cells in which diseases can be studied and on which drugs can be tested.
Though much of the focus – and debate – of stem cell research has centered on embryonic stem cells, researchers have also forged ahead with investigations of specialized adult stem cells. These cells, though limited to producing particular types of tissue, show promise in treating diseases of those tissues and in helping scientists understand the basic biology behind their functioning.
In labs across the country and around the world, scientists have begun unraveling the stem cell’s secrets in a scientific effort that has some researchers predicting the 21st century will be the century of the cell, much as the 20th was the century of the gene.
The Harvard Stem Cell Institute aims to speed the efforts of researchers at Harvard through intellectual, logistical, financial, and moral support. The institute seeks to encourage the basic science and the practical treatments that could ease the lives of 150 million people across the country suffering five kinds of tissue or organ failure.
Those diseases include diabetes, where insulin-producing beta cells are destroyed; neurodegenerative diseases, such as Parkinson’s disease, where midbrain neurons are damaged; blood diseases, such as leukemia, where abnormal blood cells are produced; cardiovascular disease, where heart muscle tissue is damaged in heart attacks; and musculoskeletal diseases such as muscular dystrophy, where bone and muscle are damaged.
Some avenues of inquiry
Researchers will move ahead on multiple fronts designed to address all five of
the disease types in a comprehensive effort that the institute’s organizers expect to be the largest and broadest ever mounted.
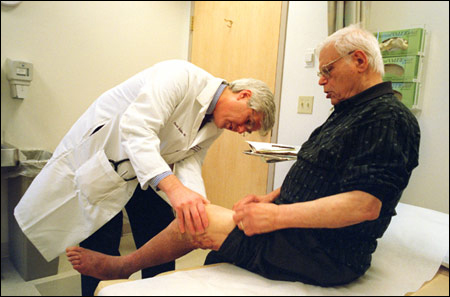
Institute Co-Director David Scadden, a professor of medicine at Harvard Medical School, said one avenue of inquiry among many will examine the stem cells’ environment to see what influence it has on the cell.
Scadden said it is likely that the stem cells are affected by the tissues around them and that those tissues may provide clues to stem cell regulation. It has already been shown, he said, that targeting bone cells with a hormone used to treat osteoporosis results in an increase in blood stem cells in the bone marrow.
Other research uses stem cells as a model to examine cancers, Scadden said. Stem cells are relatively rare in normal tissues, with just one stem cell out of up to a million blood, muscle, or other tissue cells. Researchers increasingly believe that cancerous tumors share similar characteristics, with a few cells responsible for tumor growth.
If that’s the case, Scadden said, then treatments should be designed to target the few cells responsible for growth rather than the masses of cancer cells.
“If it’s true, it really changes our thinking on cancer,” Scadden said.
Stem cells are being examined as a way to treat a host of diseases, but they may be most promising in the treatment of blood diseases, according to George Daley, an associate professor of biological chemistry and molecular pharmacology at Harvard Medical School and a physician at Children’s Hospital.
For more than a decade, blood stem cells have been given to patients recovering from chemotherapy, which can destroy the blood-producing cells in the bone marrow. The treatment allows patients to recover sooner and more fully from the chemotherapy used in certain cases of leukemia and lymphoma.
Stem cells, combined with somatic cell nuclear transfer and gene therapy, are promising in the treatment of several genetic diseases, such as severe combined immune deficiency (SCID), Daley said. SCID is a genetic abnormality in which patients lack the immune function that allows their body to fight off infection.
Current treatments for the disease involve bone marrow transplantation, but finding a perfect tissue match is extremely difficult, Daley said. Gene therapy alone has proven successful against SCID, Daley said, but the virus used to deliver the genes has been shown to cause leukemia.
Stem cell researchers hope to take a multipronged approach, using somatic cell nuclear transfer to create blood stem cells that are a match for the SCID patient and then using gene therapy to correct the patient’s original genetic abnormality that led to SCID. The cells would be injected into the SCID patient and strengthen his or her immune system.
Connecting researchers
The techniques used in treating SCID would be useful in a wide variety of diseases requiring tissue replacement. One of the institute’s strengths will be its ability to connect researchers investigating different diseases but grappling with similar problems. Today these researchers often toil in laboratories or institutes dedicated to a particular disease, apart from other stem cell researchers working on other diseases.
With the institute, researchers working on diabetes struggling to get a stem cell to develop into an insulin-producing beta cell, for example, will be able to compare notes with researchers working on Parkinson’s disease struggling to get a stem cell to develop into a neuron.
By finding common themes among different diseases, researchers will be able to share insights and collaborate in solving common problems. Institute Co-Director Douglas Melton, Thomas Dudley Cabot Professor of the Natural Sciences and investigator for the Howard Hughes Medical Institute, identified common problems that will be faced by all stem cell researchers.
Those problems include isolating stem cells, growing them efficiently, getting stem cells to differentiate into specific tissues, and transplanting and incorporating the new cells into the right location in the host body.
To further researchers’ efforts, the institute will develop new core facilities, animal models, technologies, and reagents badly needed now and critical to the advancement of stem cell knowledge.
The newly developed resources will enable investigators to sort cells, to see stem cells interacting with their environment, and to transfer nuclei between cells. Researchers also need a way to determine whether stem cells are developing into particular types of tissue cells, such as muscle or skin, in a process called differentiation.
Once developed, these resources will help the Harvard Stem Cell Institute mount a coordinated campaign to understand stem cells and how they can be used to treat disease.
“There’s going to be lots of things going on in parallel,” Scadden said.




